Infarmed approves marketing of three cannabis products

Infarmed has approved three authorizations to place cannabis-based preparations on the market for medicinal purposes.
Anvisa joins the OAS Program – Full integration

Anvisa approved a joint ordinance with the Brazilian Federal Revenue Service, allowing it to participate in the Integrated AEO Program.
Anvisa – Innovative new regulations for the food sector

Anvisa has published the new regulatory framework for the regularization of food in the context of the National Health Surveillance System.
Anvisa announces selection of innovative medical devices

visa chose ten innovative projects for the pilot project, which were announced at the Technological Advances in Medical Devices Seminar.
Anvisa to update regulations on tartrazine on labels

Anvisa will be forced to change a rule that will require the presence of the yellow tartrazine dye to be clearly indicated on food labels.
FDA launches ingredients directory for food supplements

The US FDA has updated its ingredients directory to include information on dietary supplements and other substances.
Anvisa publishes GGMED Management Report

The record number of approvals shows the Agency’s progress in terms of efficiency and commitment to generics and innovation policies.
Begins biological task force on Optimized Analysis Project

Anvisa starts a new phase of the Online Optimized Analysis Project to speed up post-registration biological applications.
Anvisa publishes sunscreen regulation manual

Anvisa (National Health Surveillance Agency) has released a guide containing crucial information for registering sunscreens.
Medicines and clinical research approved in 2023
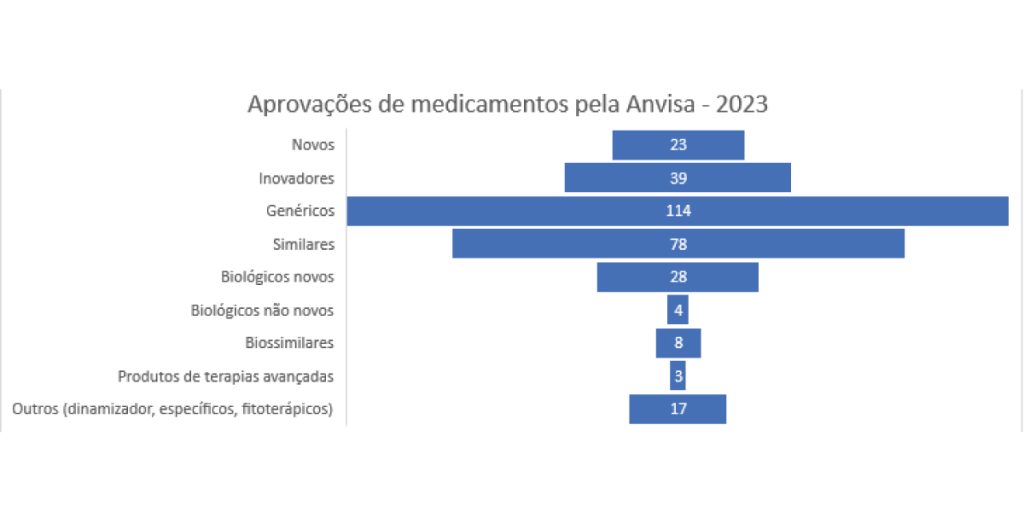
In 2023, Anvisa (National Health Surveillance Agency) accelerated approvals, modernized regulations and increased regulatory publications.
